

Licensed professionals can receive the latest Listerine clinical information and patient resources.

A TRUE ORIGINAL WITH MORE THAN A CENTURY OF ANTISEPTIC POWER
LISTERINE® Antiseptic has been a powerful germ killer for more than 140 years.

Inspired by DR Lister, Dr Lawrence created LISTERINE—for use as an antiseptic in surgery and for disinfecting wounds—in 1879.

By 1885, LISTERINE® Antiseptic was beginning to be used by dental professionals. Although LISTERINE® Antiseptic has been formulated with the same active ingredients since the 1879 and has been a household name for well over a century, over the past four decades we have also gained extensive clinical evidence of its safety and efficacy supporting its use.
THE UNIQUE INGREDIENTS BEHIND THE GERM-KILLING POWER OF LISTERINE ® ANTISEPTIC
The alcohol in LISTERINE® Antiseptic only acts as a solvent that delivers the active ingredients, which are the essential oils. It is the essential oils that do the work. The essential oils were inspired by the antimicrobial properties of the oils found from plants.1,2
Essential oils in LISTERINE® Antiseptic
LISTERINE® ANTISEPTIC IS THE MOST EXTENSIVELY TESTED OTC MOUTHWASH

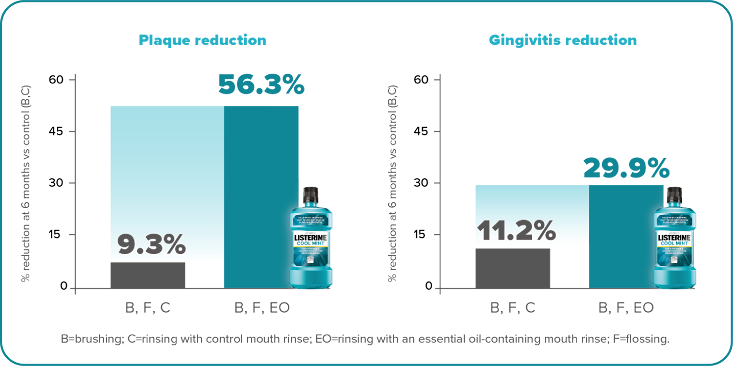
A pivotal 6-month clinical study by Sharma et al in 2004 demonstrated that brushing and flossing reduced whole-mouth plaque by only 9.3% and gingivitis by 11.2% compared to brushing alone. But when LISTERINE® Antiseptic rinse was added to brushing and flossing, whole-mouth plaque was reduced by 56.3% and gingivitis by 29.9%. 4
Adding LISTERINE® Antiseptic to brushing and flossing led to greater reductions in whole-mouth plaque and gingivitis4
In 2015, in the most robust collection of clinical data of its kind, one landmark meta-analysis of 29 studies based its analyses on site-specific plaque and gingivitis scores. This metanalysis showed LISTERINE® Antiseptic, through the action of plaque reduction, helps restore gingival tissue to health, resulting in 2 times as many gingival health sites as mechanical methods alone.14
PROVEN SAFETY AND TOLERABILITY OF LISTERINE® IN CLINICAL STUDIES
Because LISTERINE® Antiseptic is so effective at killing bacteria, some dental professionals may wonder if its potency could result in oral adverse events or contribute to antimicrobial resistance. As powerful as LISTERINE® is, its safety is supported by fifteen 6-month studies conducted over a 20-year period in 3203 subjects.8+ Data indicate that treatment-related oral adverse events reported in LISTERINE® groups were low (<1.0%) and generally similar to those reported in brushing-only groups. No serious treatment-related adverse events were reported in these studies.8
- Well tolerated on oral mucosa 8
- No negative effect on balance of oral bacteria 15,16
- No detectable increase in microbial resistance 15,16
- No detectable increase in opportunistic pathogens 15,16
- No negative impact detected due to alcohol 17-22
- Compatible with composite restorations and dental implants 23-24

*Includes a variety of LISTERINE® rinses, including alcohol- and nonalcohol-containing formulations.
References: 1. Seow YX, Yeo CR, Chung HL, Yuk H-G. Plant essential oils as active antimicrobial agents. Crit Rev Food Sci Nutr. 2014;54(5):625-644 . 2. Foster JS, Pan PC, Kolenbrander PE. Effects of antimicrobial agents on oral biofilms in a saliva-conditioned flowcell. Biofilms. 2004;1:1-10. 3. DePaola LG, Spolarich AE. Safety and efficacy of antimicrobial mouthrinses in clinical practice. J Dent Hygiene. 2007;81(special suppl):13-25. 4. Sharma N, Charles CH, Lynch MC, et al. Adjunctive benefit of an essential oil-containing mouthrinse in reducing plaque and gingivitis in patients who brush and floss regularly: a six-month study. J Am Dent Assoc. 2004;135(4):496-504. 5. Fine DH, Furgang D, Lieb R, Korik I, Vincent JW, Barnett ML. Effects of sublethal exposure to an antiseptic mouthrinse representative plaque bacteria. J Clin Periodontol. 1996;23(5):444-451. 6. Ricci-Nittel D, Fourre T. In vivo evaluation of antimicrobial activity of an essential-oil mouthrinse. Presented at: General Session of the International Association for Dental Research; March 16-19, 2011; San Diego, CA. Oral Session. 7. Pan PC, Harper S, Ricci-Nittel D, Lux R, Shi W. In-vitro evidence for efficacy of antimicrobial mouthrinses. J Dent. 2010;38(suppl 1):S16-S20. 8. Data on file, Johnson & Johnson Consumer Inc. 9. Gunsolley JC. A meta-analysis of six-month studies of antiplaque and antigingivitis agents. J Am Dent Assoc. 2006;137(12):1649-1657. 10. Gunsolley JC. Clinical efficacy of antimicrobial mouthrinses. J Dent. 2010;38(suppl 1):S6-S10. 11. Van Leeuwen MP, Slot DE, Van der Weijden GA. Essential oils compared with respect to plaque and parameters of gingival inflammation: a systematic review. J Periodontol. 2011;82(2):174-194. 12. Swango PA. Regular use of antimicrobial mouthrinses can effectively augment the benefits of oral prophylaxis and oral hygiene instructions at 6-month recall intervals in reducing the occurrence of dental plaque and gingivitis. J Evid Based Dent Pract. 2012;12(2):87-89. 13. Boyle P, Koechlin A, Autier P. Mouthwash use and the prevention of plaque, gingivitis and caries. Oral Dis. 2014;20(suppl 1):1-68. 14. Araujo MW, Charles CA, Weinstein RB, et al. Meta-analysis of the effect of an essential oil-containing mouthrinse on gingivitis and plaque. J Am Dent Assoc. 2015;146(8):610-622 and/or post-hoc analyses of data. 15. Minah GE, DePaola LG, Overholser CD, et al. Effects of 6 months use of an antiseptic mouthrinse on supragingival dental plaque microflora. J Clin Periodontol. 1989;16:347-352. 16. Walker C, Clark W, Tyler K, Ross N, Dills S. Evaluation of microbial shifts following long-term antiseptic mouthrinse use [abstract]. J Dent Res. 1989;68:412. Abstract 1845. 17. Aceves Argemí R, González Navarro B, Ochoa García-Seisdedos P, Estrugo Devesa A, López-López J. Mouthwash with alcohol and oral carcinogenesis: systematic review and meta-analysis. J Evid Based Dent Pract. 2020;20(2):101407. 18. International Agency For Research on Cancer (IARC). IARC monographs on the evaluation of carcinogenic risks to humans: Report of the advisory group to recommend priorities for IARC Monographs during 2015-2019. https://monographs.iarc.fr/wp-content/uploads/2018/08/14-002.pdf. Accessed May 12, 2020. 19. Food and Drug Administration. Oral health care drug products for over-the-counter human use; antigingivitis/antiplaque drug products; establishment of a monograph; proposed rules. Part Ill. Federal Register 2003;68(103):32232-32287. 20. Cole P, Rodu B, Mathisen A. Alcohol-containing mouthwash and oropharyngeal cancer: a review of the epidemiology. J Am Dent Assoc. 2003;134:1079-1087. 21. La Vecchia C. Mouthwash and oral cancer risk: an update. Oral Oncol. 2009;45:198-200. 22. Boyle P, Gandini S, Boffetta P, Negri E, La Vecchia C. Mouthwash use and oral cancer risk: quantitative meta-analysis of epidemiologic studies. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(6):e130. 23. Pelino JEP, Passero A, Martin AA, Charles CA. In vitro effects of alcohol-containing mouthwashes on human enamel and restorative materials. Braz Oral Res. 2018;32:e25. 24. Ciancio SG, Lauciello F, Shibly O, Vitello M, Mather M. The effect of an antiseptic mouthrinse on implant maintenance; plaque and peri-implant gingival tissues. J Periodontol. 1995;66(11):962-965.
Read about the unique fixed combination that makes LISTERINE® Antiseptic stand apart.
LISTERINE® Antiseptic
<p>Site-specific benefit of adding LISTERINE® Antiseptic to an oral care regimen</p>


All Fields required, unless otherwise indicated
Personal Information
Step 1
Will be used as your user name
By submitting your information above, you agree that the information you provide will be governed by our site's Privacy Policy.